This exam is to be worked on independently. You may not discuss any aspect of this exam with anyone except Professor Martin - not other students, not other faculty, no one.
Question 1
a) (10 points) Examine the interface between the two subunits. Describe the chemical nature of this interface and what forces you think drive and/or are important in subunit-subunit interactions..
Answer: Show me the . Note the number of water molecules (pink) at the interferface. This is unusual. There are some hydrophobic contacts (green), and they are certainly expected to help drive association, but not as much as at some protein-protein interfaces. Water-mediated H-bonds between amino acids on opposite sides of the interface are expected to be less precise than more direct contacts. One might expect this interface to be more "loose" than some.
b) (10 points) Looking at the location of amino acids 83 to 92, do you expect this stretch to be polar, nonpolar, or amphipathic? Is your prediction upheld? Explain your prediction.
c) (10 points) Looking at the location of amino acids 98 to 104, do you expect this stretch to be polar, nonpolar, or amphipathic? Is your prediction upheld? Explain your prediction..
Answers: Show me the .
b) Helix 83-92 is predicted to be (and is) amphiphilic, since it lies at the surface of the protein. Amino acids facing solvent are polar and residues facing the hydrophobic interior are nonpolar. Sequence is Ser-Leu-Tyr-Glu-Val-Leu-Glu-Ala-Ile-Lys -- the center is a mix of hydrophobic and polar.
c) Strand 98-104 is buried within the protein and so is expected to be uniformly nonpolar. It is, except at the ends, which are near the protein surface. Sequence is Gln-Leu-Trp-Phe-Leu-Val-Arg -- the center is uniformly hydrophobic.
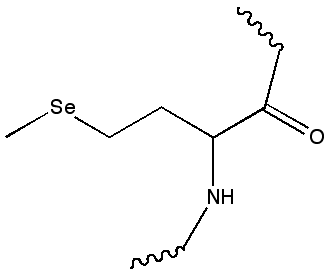
d) (10 points) This structure contains heteroatoms. Draw the chemical (structural) formula for the heteroatom MSE in this structure, using classic organic structure notation.
Answer: This protein is clearly composed of , chains A and B. But you can also recognize that each subunit is composed of , each with an independent . However, the construction is complicated in that the cores are not simply built from one contiguous chain in each.
e) (5 points) This structure contains heteroatoms. Draw the chemical (structural) formula for the heteroatom MSE in this structure, using classic organic structure notation.
in on it
Answer: This is selenomethionine, incorporated as an amino acid in the protein. That it is not a separate molecule can be deduced by the rest of the backbone. But also looking at the coord struct of MSE alone was a tip - it was not a complete amino acid (look at what should be the - it's not a carboxylate).