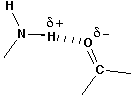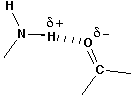B-Form DNA
This is an interactive tutorial on DNA. Read the text and on buttons or highlighted words. If you "click and drag" in the window at right, you can rotate the molecule yourself. In fact, you can do this at any time. Click on the "?" in the menu above for more tips on manipulating the molecule at right.
Click "reset" above, to return the view to the starting point.
The structure at right, B-Form duplex DNA, contains two interacting molecules: two complementary strands of DNA.
Duplex DNA is a long polymeric species made up of two individual molecules. Each strand of DNA is made of repeating units, called nucleotides. Complementary strands of DNA have nucleotides which interact favorably via base pairing. The shape and hydrogen bonding between bases ensures complementarity and their planarity allow them to "stack" on each other. But other forces are important in maintaining the structure.
The stick representation above is the actual structure of a very short stretch of DNA
Nucleotide
A nucleotide is composed of 3 parts:
The sugar rings are connected by phosphate linkages. Together these serve as the backbone of DNA.
Real DNA
Those of you who have seen textbook pictures of DNA might notice something unusual about this DNA. In fact, it is the textbooks which are unusual.
In the simple world of textbooks, the base pairs are completely "co-planar." In the real world, small, but significant forces act to distort the base pairs slightly, to achieve a real-world energy minimum.
DNA is a polymer
A is any chemical species made up of repeating units.
contains two polymeric "chains"
The units are connected together via phosphate linkages
Show a polymer with .
The "unit" in this case can be any one of four nucleotides.
Most chemical polymers are a distribution of lengths and compositions. In contrast, nature's polymer, DNA, is put together with extreme precision. This is important, since the order of the nucleotides contains information, in much the same way that the order of 0's and 1's on a computer tape contains information.
Base Pairing
Complementary refers to the way two individual bases can interact in a "lock and key" fashion. An example is the base pair between cytosine (C) and guanosine (G):
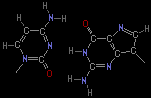
Show me a
Note that in DNA crystal structures, hydrogens (H) are not detected (why?) and so are not shown in the structure at right. Compare the structure above with that at right.
Consequently, chemists and biologists who look at these structures have to figure out where the hydrogens are placed based on their knowledge of chemistry.
Knowledge of where the H's are is important in assessing the hydrogen bonding characteristic of a base pair. Note that some of the nitrogens have lone pairs (again you have to use your knowledge of chemistry to assess where the lone pairs are placed).
Hilight a .
Hydrogen Bonding
The lone pair can hydrogen bond to the hydrogen from the opposite base, helping to stabilize the DNA.
Show me a
Show me
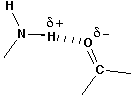
Remember that we can draw resonance structures to get a feel for charge distribution within a molecule. If we do this for the two bases, we find that the charges make for an attractive hydrogen bond.
Orbital Hybridization
Note also that all of the atoms in the base rings are sp2 hybridized (this is why the rings are planar). Click in the window and rotate the base pair.
for me. buggy
Go back to the to see the stacked planes.
Forces
Most people think that the hydrogen bonds are what hold DNA together. While they are important for the overall stability, other forces actually drive the two strands to form a helix.
Do you remember why oil forms droplets in water? Or why lipids form bilayers in water?
Nonpolar molecules want to phase separate from water.
The base part of a DNA monomer has some nonpolar ("oily") character (does it look a bit like benzene?).
Hilight in a DNA duplex. Note that phosphates are typically shown in yellow and the bound oxygens in red. Together they form a phosphate group.
Notice that the phosphates are negatively charged. Charged groups want to interact with the water dipoles and so want to be out in the water as much as possible. The bases and sugars (at least parts of them), on the other hand, are a bit "oily" and want to be away from water. The DNA duplex achieves this!
Show me a .
space filling view.
Rotate
Show me a .
Views
Chemists usually represent molecules in a "stick" view. This is equivalent to viewing a person as his or her skeleton. In reality, the electrons associated with this molecule fill space.
Show me a view.
Show me a view.
Return to the .
Furanose Sugar
the sugar component of a nucleotide.
The sugar ring is .
Why no hydrogens?
In x-ray crystallography, x-rays scatter off of the electron clouds which surround nuclei. So it is actually the "electron density" which is measured. Each hydrogen atom has one electron around its nucleus. In constrast, carbon has six electrons and phosphorous has 15 electrons. Consequently, carbon, nitrogen, oxygen, and phosphorous give much stronger signals than does hydrogen. The hydrogens are simply not detected.